Abstract 03
Determinants of sham response in tDCS depression trials: a systematic review and meta-analysis
Stefanie De Smet 3,4; Stevan Nikolin 6,7; Adriano Moffa 6,7; Paulo Suen 1; Marie-Anne Vanderhasselt 3,4,5; André R. Brunoni 1,2; Laís B. Razza 1.
1 Service of Interdisciplinary Neuromodulation, Neuroscience Laboratory (LIM-27), Department and Institute de Psychiatry, Clinical Hospital, University of São Paulo Medical School, São Paulo, Brazil;
2 Department of Internal Medicine, University of São Paulo Medical School & University Hospital, University of São Paulo, Av. Prof Lineu Prestes 2565, 05508-000, São Paulo, Brazil;
3 Department of Head and Skin, Psychiatry and Medical Psychology, Ghent University Hospital, Ghent University, Ghent, Belgium;
4 Ghent Experimental Psychiatry (GHEP) lab, Ghent, Belgium;
5 Department of Experimental Clinical and Health Psychology, Ghent University, Ghent, Belgium;
6 School of Psychiatry, University of New South Wales, Sydney, Australia; 7Black Dog Institute, Sydney, Australia.
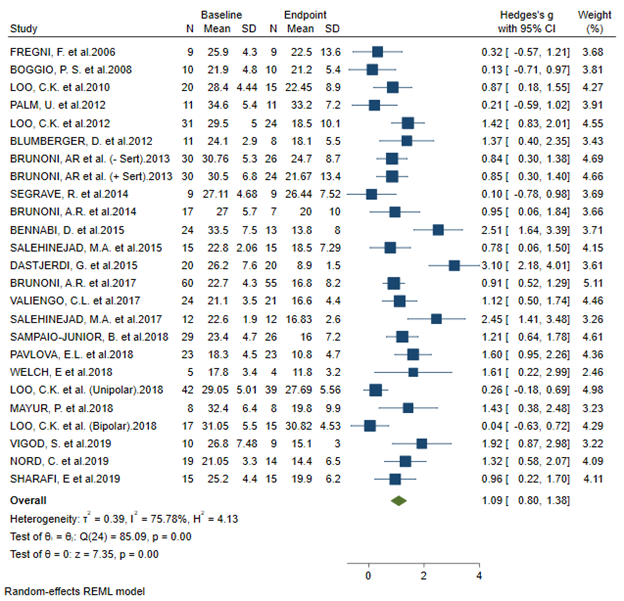
BACKGROUND: Randomised clinical trials (RCTs) investigating transcranial direct current stimulation (tDCS) efficacy for depression show significant heterogeneity in outcomes. Objective: To investigate the magnitude of the sham tDCS response and its potential moderators in the treatment of depression.
METHODS: A systematic review and aggregate meta-analysis. The systematic review was conducted in the PubMed, Scopus (EMBASE) and Cochrane Library databases. Only RCTs enrolling adult subjects with an acute depressive episode with a sham tDCS group were included.
RESULTS: Twenty-three studies (twenty-five datasets, 501 participants) were included. Sham tDCS response was large (Hedges’ g = 1.09; 95%CI: 0.8; 1.38). Secondary and subgroup analyses showed that sham protocols employing a ramp-up/ramp-down at the beginning and end of stimulation presented a significantly reduced sham response compared to other protocols. Univariate meta-regression analyses found that sham response was associated with higher risk of blinding bias, and with treatment effect size in the active tDCS group. Subgroup analyses also showed that placement of the cathode over the lateral right frontal area (F8) presented a significantly low sham response. Other moderators, including treatment resistance, baseline severity of depressive symptoms, and total charge delivered were not associated with the magnitude of the sham response.
CONCLUSIONS: The sham tDCS response was large. Our findings demonstrate the need for standardization of sham tDCS protocols and bring attention to important considerations that can guide future RCTs employing tDCS for the treatment of MDD.
FIGURE:
KEYWORDS: Transcranial direct current stimulation (tDCS); sham response; Major Depressive Disorder; placebo; Meta-analysis
ACKNOWLEDGMENTS: ARB receives grants from the National Council for Scientific and Technological Development (CNPQ, PQ-1B, to Dr. Brunoni), the Program of Academic Productivity (PIPA) of the University of São Paulo Medical School (to Dr. Brunoni). ARB is chief medical advisor of Flow ™ and has a small equity of the company. The LIM-27 laboratory of neuroscience receives grants from the Associação Beneficente Alzira Denise Hertzog da Silva. LBR is supported by São Paulo Research Foundation (Grant Number: 2019/07256-7). MAV & SDS received funding by the FWO Grant G0F4619N.
FUNDING/FINANCIAL SUPPORT: None
